Diagnostic accuracy of Juvenile Idiopathic Arthritis (JIA). Validation, refinement and exploration of new imaging techniques, followed by a randomized clinical trial (RCT) to examine the effect of intraarticular injections to the temporomandibular joints (TMJs). Oral and bone health
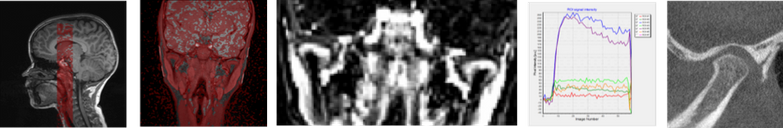
Juvenile Idiopathic Arthritis (JIA) is a heterogeneous condition including all forms of chronic arthritis of unknown origin and affects around 1-2 in 1000 children under the age of 16 (1, 2). Some speculate that oral health may play a causative role as peptides from both bacteria and virus may trigger an autoimmune response (3). Further, that inflammation of the gingiva may cause progression of the disease. There is increasing evidence that many, if not most, children with JIA will have a chronic disease with ongoing activity into adulthood, and that the temporomandibular joints (TMJs) are more frequently involved than previously believed; in up to 70% when based on MRI (4). However, the reported incidences are highly inconsistent, varying from 17% to 87%, reflecting methodological difficulties. Moreover, recent research suggests that a therapeutic window of opportunity during early disease may exist and that early initiation of therapy may be associated with slower progression of joint damage and higher rates of remission. This underscores the need for new, sensitive disease markers as many of the existing, based on clinical, laboratory and conventional imaging, are imprecise and inaccurate.
Using a cross-disciplinary (radiologists, rheumatologists, dentists, maxillofacial surgeons, physicists, mathematicians, radiographers), multinational, multicenter, longitudinal prospective design, we aim to combine clinical, laboratory and radiological data from a large number of children with JIA, to validate, refine and further explore different imaging techniques used for the assessment of JIA, with a focus on advanced MRI techniques and image analysis.
Objectives
This project addresses the current lack of imaging markers for JIA through developing and exploring precise and valid, child-specific imaging biomarkers and scoring systems allowing for evidence-based clinical practice as well as for robust drug trials. More specifically, we will
1.Develop MRI-based normal standards for the TMJ and hip joints
2.Create and validate (precision) MRI markers for active and chronic JIA disease of the wrists, hips and TMJs. Identify new, perfusion based MRI markers for active disease
3.Establish novel MRI-markers for active involvement of the TMJs including perfusion markers, and cone beam computed tomography (CBCT) based markers for chronic involvement
4.Based on the above, create and validate (clinical validity and prediction of long term outcomes) MRI scoring systems for active and chronic disease of the TMJs
5.Create prognostic, multi parametric markers for disease course / outcome of TMJ disease
6.Report the frequency of TMJ involvement in children with JIA based on MRI and CBCT
7.Investigate associations of active and chronic JIA as defined in 2 and 3 above, in univariate and multivariate models, with height, weight and body mass index, clinically assessed joint mobility and measures of disease activity and severity, oral health and bone health.
8.Examine the efficacy and safety of intraarticular injections (IAIs) to the TMJs
253 Terry Francois Street, San Francisco, CA 74059 | 1-800-000-0000
Copyright @ All Rights Reserved